Technologies and range of methods at IIB e.V.
In conjunction with the University of Rostock, the IIB has access to a very wide range of equipment, a range that has been further extended through investments funded mainly by the Mecklenburg-Vorpommern government. The research infrastructure, physical equipment, technology platforms and the associated process technologies that have been built up as a priority in the course of the research work we have carried out and that have been further developed in view of their importance for the regional economy, include the following fields in particular:
Clean room laboratory
We have a clean room laboratory conforming to Class 8 according to ISO 14644, especially for the development and production of implant surface coatings capable of releasing pharmaceuticals, which are often referred to as drug-eluting implant surfaces, and for carrying out selected production processes and product and materials testing under clean room conditions.
GLP test laboratory for biomaterials conforming to ISO 10993 at the University of Rostock
This laboratory provides a range of in vitro biocompatibility assays and in vivo tests using small animal models to assess the following parameters: cytotoxicity | haemocompatibility | pyrogenity | sensibilisation | irritation | long-term implantation | subchronic toxicity | immune reaction | biodegradation
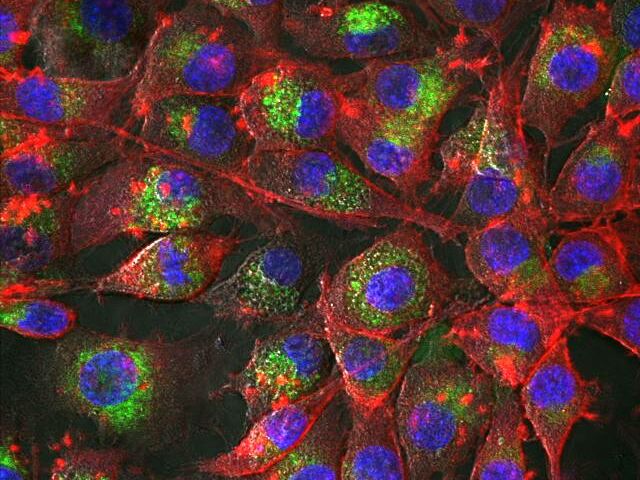
Analysis of micro and nano structures
One emphasis in our technical equipment is centred on identifying the morphological properties of biomaterials and implant surfaces, as well as of other specialised materials and components. In this field, the following are amongst the analytic procedures available: transmission electron microscopy (TEM) | scanning electron microscopy (SEM/ESEM) | confocal laser scanning microscopy (CLSM) | atomic force microscopy (AFM)) | micro computed tomography (µ-CT) | preparation techniques
Chemical and biological analysis
The following analytical procedures and laboratory equipment were set up for a wide variety of applications: high-performance liquid chromatography (HPLC) | gel permeation chromatography (GPC) | infrared spectroscopy and microscopy (FT-IR) | UV-Vis photometry using nanocells | quantitative PCR | polyacrylamide gel electrophoresis | agarose gel electrophoresis | local control assays | microplate viewers for absorption, fluorescence and luminescence | confocal laser scanning microscopy (CLSM) | cell culture laboratory | cooled centrifugation | ultrasound homogenisation
Biomechanical and physio-chemical materials testing
The following test procedures can be used for the multi-factorial identification of materials properties: tensile, pressure, bending and fatigue tests | acclimatised tests | use of physiological testing media in liquid cells | dynamic mechanical analysis (DMA) | dielectric analysis (DEA) | differential scanning calorimetry (DSC) | rheometry and viscometry | measurement of contact angle
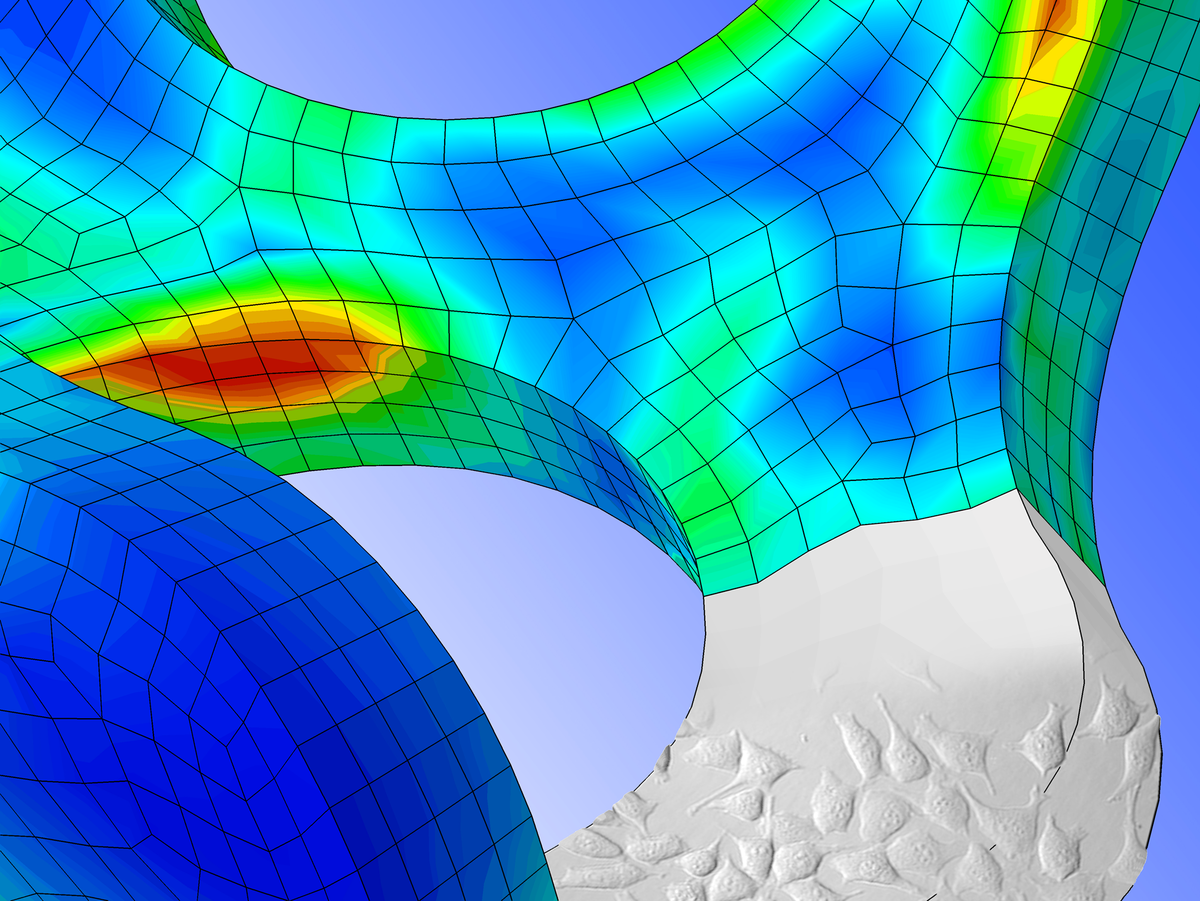
Implant technology
Design optimisation, simulation, prototype production as well as materials testing and in vitro functional testing are particularly addressed in our established implant technologies. A range of high-performance computer aided engineering (CAE) tools are available for structural design (using CAD) and manufacturing (using CAM), as well as for the simulation of implants as they interact with biological tissue using finite element analysis (FEA). Our wide range of services is rounded out by the technical documentation service for the developments that we provide, right up to drawing up the necessary manufacturing documentation.